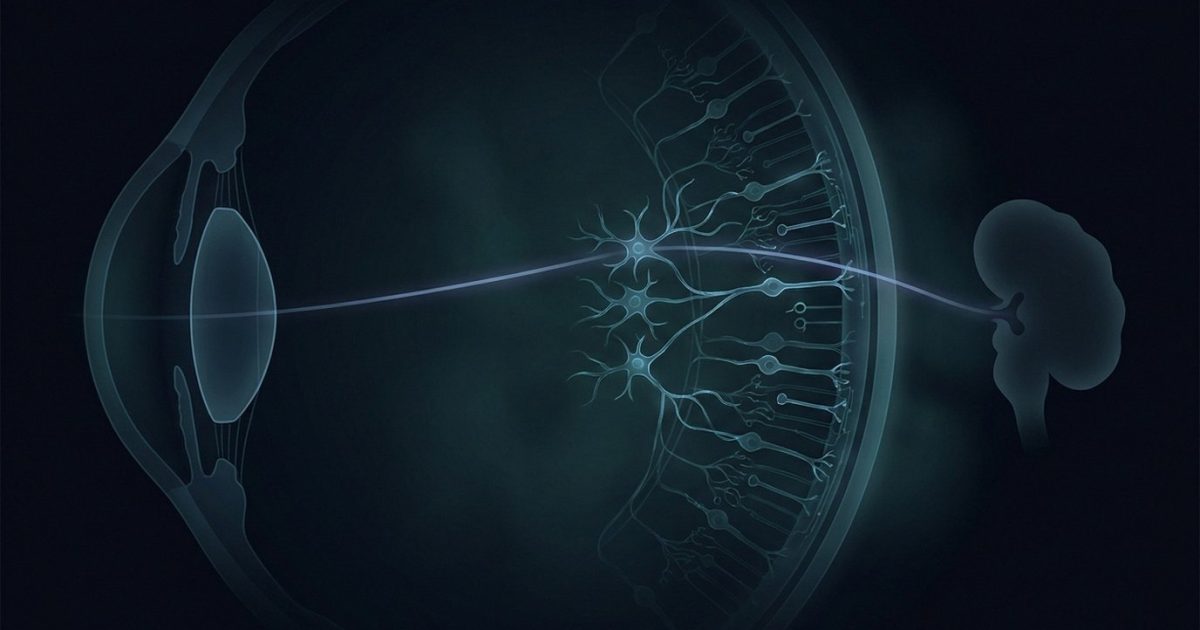
A photon enters the eye. Not a rod. Not a cone. A third class of retinal cell — the intrinsically photosensitive retinal ganglion cell, or ipRGC — absorbs it via the photopigment melanopsin, whose sensitivity peaks in the 446–477 nm blue-cyan band. That signal travels the retinohypothalamic tract to the suprachiasmatic nucleus, relayed onward to the pineal gland. The pineal stops making melatonin. Eight lux at 480 nm is enough. Fifteen minutes is enough. That is what artificial light at night does at the neurobiological level. For the broader health consequences, see light pollution and human health: the science of darkness, disrupted.
The Discovery of ipRGCs: Rewriting Retinal Biology
Before 2002, the textbook said rods and cones were the only light-sensitive cells in the mammalian retina. Two papers published on the same day ended that consensus.
David Berson, Felice Dunn, and Motoharu Takao at Brown University directly recorded from individual retinal ganglion cells projecting to the suprachiasmatic nucleus. Their paper — “Phototransduction by retinal ganglion cells that set the circadian clock” (Science 295:1070–1073, 2002) — showed these cells depolarised in response to light even when all synaptic input from rods and cones was pharmacologically blocked. The ganglion cells themselves were photoreceptors.
In the same issue of Science, Hattar, Liao, Takao, Berson, and Yau characterised the molecular identity of the photopigment: melanopsin (Science 295:1065–1070, 2002). Melanopsin-expressing cells mapped precisely onto the intrinsically photosensitive population. Their axons projected to the SCN, the olivary pretectal nucleus, and the intergeniculate leaflet. The photoreceptor and its downstream circuit were identified simultaneously.
Melanopsin itself had been isolated earlier by Ignacio Provencio and colleagues — in the Journal of Neuroscience, not PNAS as sometimes cited: volume 20, issue 2, pages 600–605, January 2000. They reported a novel opsin expressed in cells of the human inner retina, structurally similar to non-mammalian opsins. The 2002 papers completed the circuit Provencio’s discovery had opened.
Numbers: the adult human retina contains approximately 3,000 ipRGCs — one to three percent of all retinal ganglion cells. Sparse, but wired directly to the clock. Their coverage is sufficient to capture ambient photon flux across the full visual field, which is all circadian entrainment requires.
From Photon to Suprachiasmatic Nucleus
Melanopsin activation at 446–477 nm triggers a signalling cascade that reaches the brain’s master clock via a dedicated anatomical highway — the retinohypothalamic tract.

At the cell level, melanopsin is a G-protein-coupled opsin that uses a retinaldehyde chromophore. Photon absorption triggers a Gq/11-linked phospholipase C cascade that depolarises the ipRGC through TRPC channels. This is a slow, sustained response — suited to integrating ambient light levels over minutes rather than resolving spatial detail over milliseconds. Rods and cones also feed into ipRGCs via inner retinal circuitry, but the intrinsic melanopsin response is what the retinohypothalamic tract carries for circadian purposes.
The retinohypothalamic tract (RHT) is the monosynaptic projection from ipRGC axons to the suprachiasmatic nucleus (SCN) in the anterior hypothalamus. It uses glutamate as its primary neurotransmitter, with PACAP as co-transmitter. Glutamate release at the SCN during subjective night produces phase shifts: delays when light arrives early in the night, advances when it arrives late.
The SCN is a bilateral structure of approximately 20,000 neurons — the master circadian pacemaker. Its molecular machinery runs on CLOCK and BMAL1 proteins driving expression of Period and Cryptochrome genes, whose protein products feed back to inhibit CLOCK/BMAL1 activity in the classical ~24-hour feedback loop. Light input via the RHT resets this oscillation to the solar day. ALAN keeps the SCN in perpetual light-phase signalling.
From SCN to Pineal: How Melatonin Gets Suppressed
The SCN does not project directly to the pineal. Melatonin suppression by light is a neural command relayed through a four-step autonomic chain.
First: SCN outputs reach the paraventricular nucleus (PVN) of the hypothalamus. Second: PVN preganglionic fibres descend through the spinal cord’s intermediolateral column to the superior cervical ganglion (SCG). Third: SCG axons ascend along the carotid artery to innervate the pineal gland. Fourth: norepinephrine released from SCG terminals binds to beta-adrenergic receptors on pinealocytes, activating arylalkylamine N-acetyltransferase (AANAT).
AANAT is the rate-limiting enzyme in melatonin synthesis — the “melatonin rhythm-generating enzyme” in Klein’s characterisation (Klein et al., Recent Progress in Hormone Research, 1997, volume 53). AANAT converts serotonin to N-acetylserotonin; HIOMT then completes the conversion to melatonin. AANAT activity rises sharply at night and collapses within minutes of a light pulse — tracking exactly with measured blood melatonin.
Darkness does not passively allow melatonin production. Darkness removes the inhibitory light signal, the SCN shifts to night-phase output, the PVN–spinal–SCG chain activates, norepinephrine is released, AANAT rises, and melatonin enters the bloodstream. Secretion begins at the dim-light melatonin onset (DLMO), roughly two hours before habitual sleep onset, peaking in the early morning hours. ALAN intercepts at step one: the SCN stays in light-phase mode, the cascade stalls, AANAT stays low, melatonin is not produced.
The Numbers: Brainard’s Action Spectrum
The 2001 action spectrum study defined the precise wavelength window at which artificial light becomes a melatonin suppressor — and the threshold is lower than most people expect.
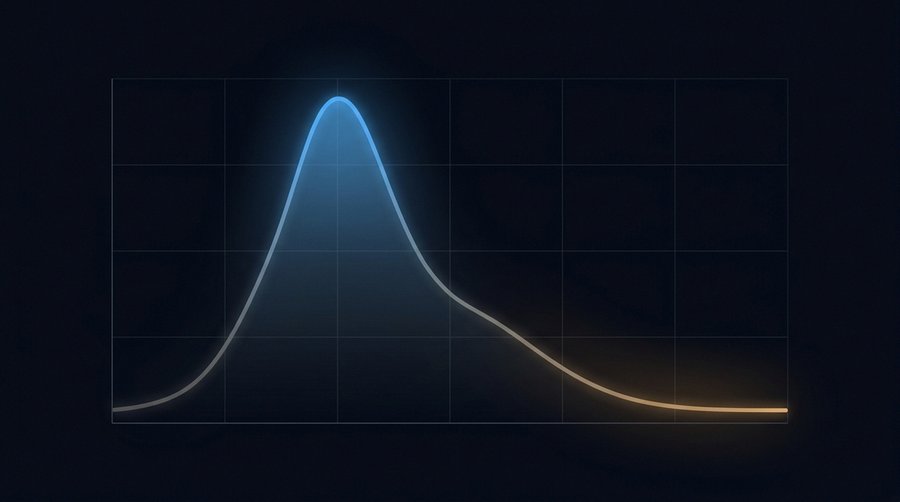
George Brainard and colleagues at Thomas Jefferson University published the definitive human action spectrum in the Journal of Neuroscience in 2001: “Action spectrum for melatonin regulation in humans: evidence for a novel circadian photoreceptor” (volume 21, issue 16, pages 6405–6412; PubMed 11487664). The study ran 627 melatonin suppression tests on 72 healthy subjects using full-field monochromatic light exposures from 2:00 to 3:30 a.m., covering 420 to 600 nm.
The action spectrum fit an opsin template with R² = 0.91. Most potent region: 446–477 nm. Distinct from rod sensitivity (~498 nm) and from S-cone and L/M-cone peaks. A single photopigment — later identified as melanopsin — appeared to drive the response. At 480 nm (within the peak band, the commonly cited rounded value), approximately 8 lux of white light is sufficient to produce measurable melatonin suppression. That is not much: roughly the horizontal illuminance two metres from a 40-watt equivalent LED, or the light level that passes under a closed door from a lit corridor.
Cajochen and colleagues at Basel confirmed the LED-specific version in 2011 (Journal of Applied Physiology 110(5):1432–1438): five hours of evening LED-backlit screen exposure — which emits substantially more 464 nm light than non-LED screens — suppressed the evening melatonin rise significantly and increased objective sleepiness. Harvard research built on Brainard’s action spectrum found blue light suppresses melatonin for roughly twice as long as green light of equal photopic intensity, shifting the circadian phase by approximately three hours versus 1.5 hours for green. The practical implication is direct: a 4,000 K LED at 8 lux has approximately the same melatonin-suppression impact as a 2,700 K source at double the lux. CCT is not an aesthetic variable. It is a dose variable.
What This Means for ALAN Research
The ipRGC pathway reframes what ALAN dose actually means: not photopic lux alone, but melanopic irradiance — the photon flux weighted to the wavelengths the clock actually uses.
Photopic lux — the standard measurement unit for light — is weighted to human visual sensitivity, peaking at 555 nm. The ipRGC system peaks at 446–477 nm. A warm 2,700 K source can be photometrically bright but spectrally low in melanopic irradiance. A visually dim 5,000 K blue-rich source can carry substantial melanopic punch. The CIE S 026 framework (2018) formalised this: it defines melanopic equivalent daylight illuminance (m-EDI), weighted to the melanopsin action spectrum, as the circadian-relevant metric.
For ALAN research using satellite data, this matters. VIIRS DNB measures panchromatic radiance — no spectral resolution, no melanopic weighting. The same satellite reading covers a 2,700 K warm street lamp and a 5,000 K cool-white LED. Their biological dose differs by a factor of three or more. Epidemiological correlations between satellite-measured sky brightness and health outcomes are thus inherently conservative estimates of actual circadian dose — and likely understate true associations for populations under high-CCT outdoor lighting.
The critical exposure window is the two hours before DLMO. This is where ipRGC sensitivity is highest and even modest melanopic doses can delay the clock by an hour or more. Indoor ALAN — screens, overhead LEDs — dominates this window. Outdoor ALAN from streetlights adds to it through curtains and windows. For street-lighting policy implications, see EN 13201 explained and the France 2018 lighting decree — the only national CCT ceiling in EU outdoor lighting law.
Implications for Lighting Practice and Nordic Populations
Warm light, screen filters, and the two-hour pre-sleep window are not lifestyle preferences — they are mechanistically grounded interventions at specific points in the ipRGC cascade.
Three intervention points follow directly from the pathway. Spectral: shift light sources below 3,000 K. A 2,700 K warm-white LED produces approximately 60% less melanopic irradiance than a 4,000 K source at equal photopic lux, because the energy in the 446–477 nm band drops sharply as CCT falls. Screen-dimming software such as f.lux or Night Shift shifts spectra toward longer wavelengths — a partial but measurable intervention in the pre-sleep period. Intensity: reduce photon flux below the 8-lux threshold in the bedroom. Temporal: protect the two hours before DLMO from blue-enriched light exposure entirely.
For Nordic populations — where in winter the natural zeitgeber signal compresses to a few minutes of twilight — every artificial light source carries disproportionate circadian weight. Stockholm at 59°N has fewer than seven hours of daylight in December. In Tromsø at 69°N, polar night eliminates direct sunlight entirely for approximately two months. The same 8-lux ALAN dose that shifts a Lisbon resident’s clock by an hour may shift a Tromsø resident’s clock by considerably more, because there is no competing daytime zeitgeber signal to counterbalance it. For the population-level consequences of this asymmetry, see Nordic chronobiology: polar night, midnight sun, and the ALAN paradox.
Melatonin as Oncostatic Agent and Therapeutic
Melatonin signals darkness. It also carries oncostatic properties — and chronic suppression by ALAN removes a brake on cellular proliferation across multiple tissue types.
Russel Reiter at the University of Texas Health Science Center has been the most systematic reviewer of melatonin’s oncostatic role, across decades of publications in the Journal of Pineal Research. The mechanisms are multiple: melatonin suppresses tumour cell proliferation via MT1 and MT2 receptor signalling; it inhibits angiogenesis; it promotes apoptosis; and it scavenges free radicals, protecting DNA during the night-phase cellular maintenance window (Reiter et al., Journal of Pineal Research, 2016). The oncostatic case for reducing ALAN is not the same as the case for melatonin supplementation — mechanistically related, epidemiologically distinct claims. The Nurses’ Health cohorts — Schernhammer et al. 2001 (JNCI: NHS I, +36% breast cancer risk for ≥30 years night shift) and 2006 (Epidemiology: NHS II, +79% for >20 years) — document the population-level consequence of chronically suppressed melatonin in high-exposure shift workers. For the IARC classification and full cancer evidence, see shift work, IARC Group 2A, and the cancer evidence.
Self-medication with melatonin supplements is not recommended here. Pharmacological doses (1–10 mg) produce supraphysiological concentrations that overwhelm receptor regulation and carry risks in specific populations. Chronobiologists work with 0.1–0.5 mg for phase-shifting purposes — closer to physiological. For the conservation biology parallel, see how ALAN disrupts wildlife circadian physiology via the same melanopsin-based mechanism conserved across vertebrates.
Frequently Asked Questions
Can a screen at night suppress my melatonin?
Yes — if used in the two hours before habitual sleep at typical brightness. A modern smartphone at full brightness emits significant melanopic irradiance in the 446–477 nm band. Cajochen et al. (2011, Journal of Applied Physiology) showed five hours of LED-backlit screen exposure suppressed the evening melatonin rise. Night-mode settings (f.lux, Night Shift) reduce the short-wavelength component; they do not eliminate it. Dimming further has an additive protective effect because ipRGC response scales with photon flux as well as wavelength.
What colour temperature is safest at night?
Below 2,700 K for any light source used in the pre-sleep window. PC-amber (approximately 1,800–2,200 K) produces the lowest melanopic irradiance per photopic lux. A 2,700 K warm-white LED produces approximately 60% less melanopic irradiance than a 4,000 K source at equal lux. France’s 2018 lighting decree mandates ≤3,000 K for outdoor luminaires nationally — the only such CCT ceiling in EU law — precisely because CCT is a biological dose variable, not cosmetic.
Does melatonin supplementation reverse light pollution effects?
Not in any clinically validated sense for healthy ALAN-exposed adults. Exogenous melatonin at 1–10 mg doses can shift circadian phase for jet lag or circadian disorders, but does not restore the full oncostatic, immune-modulating, and DNA-repair profile of endogenous physiological melatonin. The primary evidence-grounded intervention is reducing ALAN exposure in the pre-sleep window — not supplementing around it.
Are streetlights bright enough to suppress melatonin?
Depending on CCT and bedroom intrusion geometry — yes. Streetlights at 4,000 K or above penetrating through thin curtains can reach 8 lux or more at eye level. A 2,700 K lamp at the same outdoor lux produces less than half the melanopic irradiance at 480 nm. Full blackout curtains reduce photon flux below the suppression threshold regardless of CCT. The 8-lux figure from Brainard et al. 2001 is the horizontal illuminance reference; vertical illuminance at the eye depends on source angle and window geometry.